Strategic molecular design of closo-ortho-carboranyl luminophores to manifest thermally activated delayed fluorescence†
Abstract
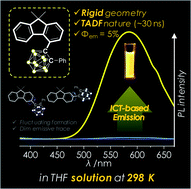
In this paper, we propose a strategic molecular design of closo-o-carborane-based donor–acceptor dyad system that exhibits thermally activated delayed fluorescence (TADF) in the solution state at ambient temperature. Planar 9,9-dimethyl-9H-fluorene-based compounds with closo- and nido-o-carborane cages appended at the C2-, C3-, and C4-positions of each fluorene moiety (closo-type: 2FC, 3FC, 4FC, and 4FCH, and nido-type: nido-4FC = [nido-form of 4FC]·[NBu4]) were prepared and characterized. The solid-state molecular structure of 4FC exhibited a significantly distorted fluorene plane, which suggests the existence of severe intramolecular steric hindrance. In photoluminescence measurements, 4FC exhibits a noticeable intramolecular charge transition (ICT)-based emission in all states (solution at 298 K and 77 K, and solid states); however, emissions by other closo-compounds were observed in only the rigid state (solution at 77 K and film). Furthermore, nido-4FC did not exhibit emissive traces in any state. These observations verify that all radiative decay processes correspond to ICT transitions triggered by closo-o-carborane, which acts as an electron acceptor. Relative energy barriers calculated by TD-DFT as dihedral angles around o-carborane cages change in closo-compounds, which indicates that the structural formation of 4FC is nearly fixed around its S0-optimized structure. This differs from that for other closo-compounds, wherein the free rotation of their o-carborane cages occurs easily at ambient temperature. Such rigidity in the structural geometry of 4FC results in ICT-based emission in solution at 298 K and enhancement of quantum efficiency and radiative decay constants compared to those for other closo-compounds. Furthermore, 4FC displays short-lived (∼0.5 ns) and long-lived (∼30 ns) PL decay components in solution at 298 K and in the film state, respectively, which can be attributed to prompt fluorescence and TADF, respectively. The calculated energy difference (ΔEST) between the first excited singlet and triplet states of the closo-compounds demonstrate that the TADF characteristic of 4FC originates from a significantly small ΔEST maintained by the rigid structural fixation around its S0-optimized structure. Furthermore, the strategic molecular design of the o-carborane-appended π-conjugated (D–A) system, which forms a rigid geometry due to severe intramolecular steric hindrance, can enhance the radiative efficiency for ICT-based emission and trigger the TADF nature.


 Please wait while we load your content...
Please wait while we load your content...