Coordinative-to-covalent transformation, isomerization dynamics, and logic gate application of dithienylethene based photochromic cages†
Abstract
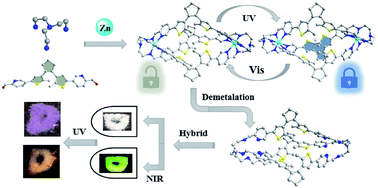
Photochromic coordinative cages containing dynamic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N imine bonds are assembled from a dithienylethene-based aldehyde and tris-amine precursors via metallo-component self-assembly. The resulting metal-templated cages are then reduced and demetalated into pure covalent-organic cages (COCs), which are otherwise difficult to prepare via de novo organic synthesis. Both the obtained coordinative and covalent cages can be readily interconverted between the ring-open (o-isomer) and one-lateral ring-closed (c-isomer) forms by UV/vis light irradiation, demonstrating distinct absorption, luminescence and photoisomerization dynamics. Specifically, the ring-closed c-COCs show a blue-shifted absorption band compared with analogous metal-templated cages, which can be applied in photoluminescence (PL) color-tuning of upconversion materials in different ways, showing potential for constructing multi-readout logic gate systems.
N imine bonds are assembled from a dithienylethene-based aldehyde and tris-amine precursors via metallo-component self-assembly. The resulting metal-templated cages are then reduced and demetalated into pure covalent-organic cages (COCs), which are otherwise difficult to prepare via de novo organic synthesis. Both the obtained coordinative and covalent cages can be readily interconverted between the ring-open (o-isomer) and one-lateral ring-closed (c-isomer) forms by UV/vis light irradiation, demonstrating distinct absorption, luminescence and photoisomerization dynamics. Specifically, the ring-closed c-COCs show a blue-shifted absorption band compared with analogous metal-templated cages, which can be applied in photoluminescence (PL) color-tuning of upconversion materials in different ways, showing potential for constructing multi-readout logic gate systems.


 Please wait while we load your content...
Please wait while we load your content...