The roles of chalcogenides in O2 protection of H2ase active sites
Abstract
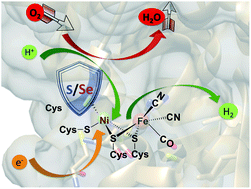
At some point, all HER (Hydrogen Evolution Reaction) catalysts, important in sustainable H2O splitting technology, will encounter O2 and O2-damage. The [NiFeSe]-H2ases and some of the [NiFeS]–H2ases, biocatalysts for reversible H2 production from protons and electrons, are exemplars of oxygen tolerant HER catalysts in nature. In the hydrogenase active sites oxygen damage may be extensive (irreversible) as it is for the [FeFe]–H2ase or moderate (reversible) for the [NiFe]–H2ases. The affinity of oxygen for sulfur, in [NiFeS]–H2ase, and selenium, in [NiFeSe]–H2ase, yielding oxygenated chalcogens results in maintenance of the core NiFe unit, and myriad observable but inactive states, which can be reductively repaired. In contrast, the [FeFe]–H2ase active site has less possibilities for chalcogen-oxygen uptake and a greater chance for O2-attack on iron. Exposure to O2 typically leads to irreversible damage. Despite the evidence of S/Se-oxygenation in the active sites of hydrogenases, there are limited reported synthetic models. This perspective will give an overview of the studies of O2 reactions with the hydrogenases and biomimetics with focus on our recent studies that compare sulfur and selenium containing synthetic analogues of the [NiFe]–H2ase active sites.


 Please wait while we load your content...
Please wait while we load your content...