Open-circuit-voltage shift of over 0.5 V in organic photovoltaic cells induced by a minor structural difference in alkyl substituents†‡
Abstract
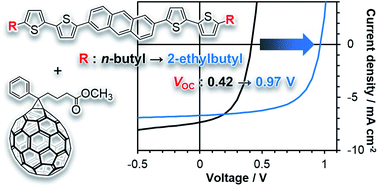
The recent surge in the efficiency of organic photovoltaic devices (OPVs) largely hinges on the reduction of energy loss (Eloss) that leads to improvements in open-circuit voltage (VOC). However, there are still many unclarified factors regarding the relationship between the molecular structure and VOC, hampering the establishment of widely applicable, effective principles for the design of active-layer materials. In this contribution, we examine the origin of the large VOC shifts induced by minor structural differences in end-alkyl substituents on a series of anthracene-based p-type compounds. The examined p-type compounds are all highly crystalline, thereby enabling detailed investigation of the molecular packing with X-ray diffraction analysis. At the same time, they are strongly aggregating and hardly soluble; therefore, they are deposited with the aid of a photoprecursor approach which we have been employing for controlled deposition of insoluble acene-based organic semiconductors. The resultant OPVs afford the highest VOC of 0.966 V when the end-alkyl groups are 2-ethylbutyl, and the lowest of 0.419 V when n-butyl is used in replacement of 2-ethylbutyl. X-ray diffraction analyses and density-functional-theory calculations indicate a critical impact of the non-slipped herringbone arrangement on the observed large loss in VOC. This type of molecular arrangement is prohibited when branched alkyl chains are introduced at the ends of linear π-systems, which we consider an important factor contributing to the relatively high VOC obtained with the 2-ethylbutyl derivative. These results may serve as a basis of useful molecular-design rules to avoid unnecessary losses in VOC.
- This article is part of the themed collections: Editor’s Choice: Malika Jeffries-EL and Papers celebrating the birthday and achievements of Professor Michael Wasielewski


 Please wait while we load your content...
Please wait while we load your content...