Abstract
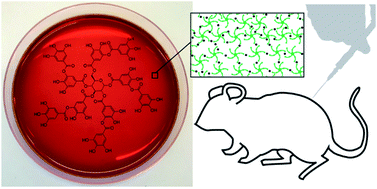
In vivo forming hydrogels are of interest for diverse biomedical applications due to their ease-of-use and minimal invasiveness and therefore high translational potential. Supramolecular hydrogels that can be assembled using metal–phenolic coordination of naturally occurring polyphenols and group IV metal ions (e.g. TiIV or ZrIV) provide a versatile and robust platform for engineering such materials. However, the in situ formation and in vivo response to this new class of materials has not yet been reported. Here, we demonstrate that metal–phenolic supramolecular gelation occurs successfully in vivo and we investigate the host response to the material over 14 weeks. The TiIV–tannic acid materials form stable gels that are well-tolerated following subcutaneous injection. Histology reveals a mild foreign body reaction, and titanium biodistribution studies show low accumulation in distal tissues. Compared to poloxamer-based hydrogels (commonly used for in vivo gelation), TiIV–tannic acid materials show a substantially improved in vitro drug release profile for the corticosteroid dexamethasone (from <1 day to >10 days). These results provide essential in vivo characterization for this new class of metal–phenolic hydrogels, and highlight their potential suitability for biomedical applications in areas such as drug delivery and regenerative medicine.


 Please wait while we load your content...
Please wait while we load your content...