Developing two-dimensional solid superacids with enhanced mass transport, extremely high acid strength and superior catalytic performance†
Abstract
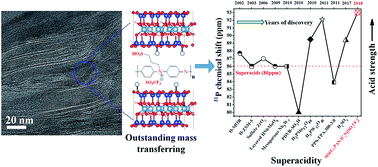
Solid acids have been widely used as heterogeneous catalysts in developing green and sustainable chemistry. However, it remains a challenge to improve the mass transport properties and acid strength of solid acids simultaneously. Herein, we report a class of two dimensional (2D) layered hybrid solid acids with outstanding mass transfer and extremely high acid strength by incorporating sulfonated polymers in-between montmorillonite layers. The 2D layered structure and broad distribution of pore sizes allow for highly efficient mass transport of substrate molecules into and out of the solid acids. The acid strength of these solid acids was found to be stronger than that of 100% H2SO4, H3PW12O40 and any other reported solid acids to date, as determined by 1H and 31P solid-state NMR. These 2D solid acids show extraordinary catalytic performance in biomass conversion to fuels, superior to that of H3PW12O40, HCl and H2SO4. Theoretical calculations and control experiments reveal that H-bond based interactions between the polymer and montmorillonite facilitate the unusually high acid strengths found in these samples.


 Please wait while we load your content...
Please wait while we load your content...