Activity-based ubiquitin-protein probes reveal target protein specificity of deubiquitinating enzymes†
Abstract
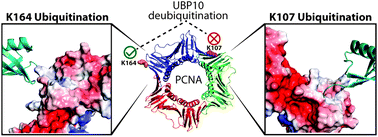
Ubiquitination is an essential eukaryotic post-translational modification that regulates various cellular processes. The removal of ubiquitin from its target protein is catalyzed by deubiquitinating enzymes (DUBs). Although it was proposed that many DUBs specifically interact and recognize ubiquitinated proteins as substrates, more direct evidence is needed to support this notion. Here we report protein-targeting activity-based DUB probes that allowed the identification of DUBs recognizing monoubiquitinated proliferating cell nuclear antigen (PCNA) in Saccharomyces cerevisiae. This new class of DUB probes contain a Michael acceptor as a warhead between ubiquitin and the target protein PCNA through a linkage that mimics the native isopeptide bond. We selected two known and biologically relevant ubiquitination sites on PCNA to generate the DUB probes. This allowed us to interrogate the site-specific deubiquitination of a target protein by DUBs. DUBs were profiled in yeast cell lysates using the two Ub-PCNA DUB probes in conjunction with two control probes that contain a noncleavable linkage but no warhead. We identified yeast DUBs through pulldown coupled with quantitative mass spectrometry analysis of the pulled down proteins. Our results showed that specific yeast DUBs recognize monoubiquitinated PCNA and corroborated previous genetic study. We also identified DUBs as potential new deubiquitinase of PCNA. Remarkably, identified DUBs clearly distinguish the different modification sites on PCNA, thus supporting a high level of DUB specificity beyond the target protein identity.


 Please wait while we load your content...
Please wait while we load your content...