UbFluor: a mechanism-based probe for HECT E3 ligases†
Abstract
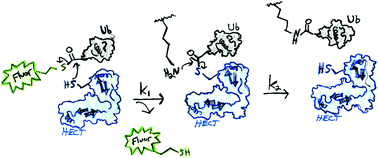
Homologues to E6AP Carboxyl Terminus E3 ubiquitin ligases (HECT, ∼28 known) are genetically implicated in cancer, neurological, hypertensive, and autoimmune disorders, and are potential drug targets to treat these diseases. The major bottleneck in the field of HECT E3s is a lack of simple assays to quantify the enzymatic activity of these enzymes in the presence of small molecules. Typical assays require E1, E2, HECT E3, ubiquitin (Ub), ATP and additional reagents to detect the resulting free poly-ubiquitin chains. To address this need, we developed UbFluor, a fluorescent thioester conjugate between the C-terminus of Ub and fluorescein-thiol (Fluor-SH). UbFluor is a mechanism-based probe that undergoes a direct transthiolation reaction with the catalytic cysteine of the model HECT E3 ligase Rsp5, producing the catalytically active Rsp5∼Ub (∼ indicates thioester) accompanied by release of Fluor-SH. The kinetics of this two-component reaction can be easily monitored with real-time fluorescence polarization (FP) assays. Importantly, UbFluor eliminates the need to use SDS-PAGE, ATP, E1 and E2 enzymes, and extra poly-ubiquitin chain detection reagents. Although the developed system lacks ATP, and E1 and E2 enzymes, we show that UbFluor can recapitulate the native ubiquitination reaction by detecting and quantifying defects in transthiolation and isopeptide ligation of Rsp5 HECT E3 alanine mutants. Based on our findings, we show that UbFluor can be utilized to conduct high-throughput screens (HTS) of small molecules against HECT ligases.

 Please wait while we load your content...
Please wait while we load your content...