Structural and reactivity insights in Mg–Zn hybrid chemistry: Zn–I exchange and Pd-catalysed cross-coupling applications of aromatic substrates†‡
Abstract
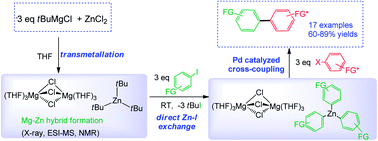
Expanding the synthetic potential of Mg–Zn hybrid organyl reagents (generated via transmetallation reactions), this study uncovers a versatile approach, involving a sequence of direct Zn–I exchange and Pd catalysed cross-coupling reactions which grants access to a wide range of asymmetric bis(aryls). By combining X-ray crystallography with ESI-MS and non-deuterium NMR spectroscopic studies, new light is shed on the heterobimetallic constitution of the intriguing organometallic species [(THF)4MgCl2Zn(tBu)Cl] (1) and [{Mg2Cl3(THF)6}+{ZntBu3}−] (2), formed through transmetallation of tBuMgCl with n equivalent amounts of ZnCl2 (n = 1 and 3 respectively). Operating by cooperative effects, alkyl-rich hybrid 2 can effectively promote direct Zn–I exchange reactions with aromatic halides in short periods of time at room temperature in THF solution. The structural elucidation of key organometallic intermediates involved in some of these Zn–I exchanges, provides new reactivity insights into how these bimetallic systems operate. Thus, while the reaction of 2 with 3 equivalents of 2-iodoanisole (3b) gives magnesium dizincate [{Mg(THF)6}2+{Zn(o-C6H4-OMe)3}2−] (4) which demonstrates the 3-fold activation of the tBu groups attached to Zn in 2, using 2-iodobenzonitrile (3i), only two tBu groups react with the substrate, affording [(THF)4MgCl(NC-o-C6H4)ZnI(o-C6H4-CN)(THF)] (7). In 7 Mg and Zn are connected by an aryl bridge, suggesting that the formation of contacted ion-pair hybrids may have a deactivating effect on the outcome of the Zn–I exchange process. A wide range of homoleptic tris(aryl) zincate intermediates have been prepared in situ and used as precursors in Pd catalysed cross-coupling reactions, affording bis(aryls) 6a–s in excellent yields under mild reaction conditions without the need of any additive or polar cosolvent such as NMP or DMI.

 Please wait while we load your content...
Please wait while we load your content...