Towards a comprehensive understanding of platinum dissolution in acidic media
Abstract
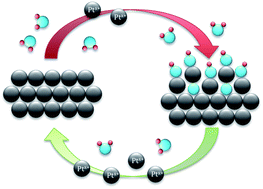
Platinum is one of the most important electrode materials for continuous electrochemical energy conversion due to its high activity and stability. The resistance of this scarce material towards dissolution is however limited under the harsh operational conditions that can occur in fuel cells or other energy conversion devices. In order to improve the understanding of dissolution of platinum, we therefore investigate this issue with an electrochemical flow cell system connected to an inductively coupled plasma mass spectrometer (ICP-MS) capable of online quantification of even small traces of dissolved elements in solution. The electrochemical data combined with the downstream analytics are used to evaluate the influence of various operational parameters on the dissolution processes in acidic electrolytes at room temperature. Platinum dissolution is a transient process, occurring during both positive- and negative-going sweeps over potentials of ca. 1.1 VRHE and depending strongly on the structure and chemistry of the formed oxide. The amount of anodically dissolved platinum is thereby strongly related to the number of low-coordinated surface sites, whereas cathodic dissolution depends on the amount of oxide formed and the timescale. Thus, a tentative mechanism for Pt dissolution is suggested based on a place exchange of oxygen atoms from surface to sub-surface positions.

 Please wait while we load your content...
Please wait while we load your content...