Economically-feasible production of a nylon monomer using RANEY® catalysts†
Abstract
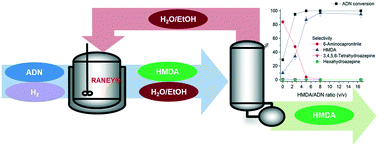
This research was aimed at developing an economically-feasible process to produce a value-added chemical used to synthesize nylon, hexamethylenediamine (HMDA), by hydrogenating adiponitrile (ADN) using an inexpensive catalyst such as RANEY® Ni or RANEY® Co. For the two RANEY®-type catalysts, the effects of reaction temperature, hydrogen pressure, catalyst/ADN ratio, and HMDA/ADN ratio in the reactant on ADN hydrogenation were investigated. As increasing H2 pressure, catalyst/ADN ratio, and HMDA/ADN ratio in the reactant, the yield of HMDA became high for both catalysts. A trend in the dependence of ADN hydrogenation on reaction temperature from 60 to 80 °C was similar for the two catalysts. Addition of HMDA to the reactant increased the HMDA yield for both catalysts. No metal leaching occurred during the hydrogenation reaction on the two catalysts. Based on the experimental results, a process simulation model was developed to determine the economic potential of the production of HMDA from ADN. Using this model, a techno-economic analysis of the experiment-based process was conducted by comparing four case studies with respect to the ratio of HMDA to ADN for each catalyst on a large scale. The process using a HMDA/ADN ratio of 8 over the RANEY® Co catalyst was the most techno-economically feasible option with the lowest minimum selling price (US$ 1.64 per kg) of HMDA produced from ADN, which is below the range of recent market prices.
- This article is part of the themed collection: Emerging Investigator Series


 Please wait while we load your content...
Please wait while we load your content...