Removal of arsenic from a denitration catalyst by reduction using methanol as a carbon source
Abstract
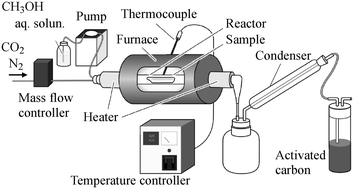
Reduction–volatilization of arsenic from a denitration catalyst obtained from a coal-fired power plant using a gaseous reducing agent was investigated to develop a dry arsenic removal process. Methanol vapour showed higher reactivity for the reduction of the arsenic species in the catalyst than solid carbon. When the catalyst was heated under a methanol atmosphere at temperatures above 350 °C, arsenic was quickly volatilized but the catalytic activity was drastically reduced. In that case, the denitration activity of the methanol-treated catalyst could be completely recovered by adding active vanadium species. It was confirmed that arsenic could be successfully removed with no decline in the catalytic activity by heating the catalyst under a methanol atmosphere in the temperature range from 250 to 275 °C.


 Please wait while we load your content...
Please wait while we load your content...