Kinetics of nanocrystal synthesis in a microfluidic reactor: theory and experiment†
Abstract
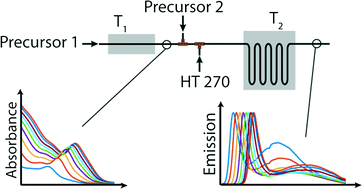
The processes occurring during nanocrystal nucleation and growth are currently not well understood. Herein, we theoretically and experimentally investigate the growth kinetics in colloidal nanocrystal synthesis. Using a novel microfluidic reactor integrating independent modules for nucleation and growth, we demonstrate the controlled, direct synthesis of high quality nanocrystals in high yield. For CdSe nanocrystals, we find that size tuning solely by variation of the reaction time and temperature does not yield product populations of optimal size dispersion or yield. Instead, we present an improved method for the synthesis of bespoke nanocrystals that relies on the controlled addition of precise amounts of additional precursor subsequent to nucleation and fine tuning of the reaction time and temperature in the second stage. Real-time spectroscopic monitoring of the produced crystals in conjunction with kinetic simulations confirms the close correspondence between the model and the experiment and elegantly quantifies the effects of temperature, concentration, additives and surfactants on conversion, growth and diffusion rates within the model framework. We show that the conversion of the precursor to a monomer follows a first order rate law and that the growth rate has a stronger temperature dependence than the conversion rate. Moreover, the surfactant concentration retards the reaction by inhibiting diffusion to the growing crystals whilst maintaining a uniform conversion rate. Finally, we demonstrate that diphenylphosphine, a common additive in CdSe synthesis, enhances the reaction rate by accelerating precursor conversion.

 Please wait while we load your content...
Please wait while we load your content...