The use of modelling to understand the mechanism of hydrogen peroxide direct synthesis from batch, semibatch and continuous reactor points of view†
Abstract
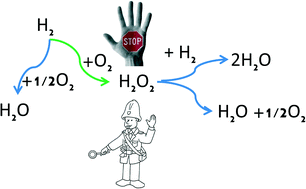
Hydrogen peroxide direct synthesis was experimentally studied in three different reactors, namely the batch, semibatch and trickle bed reactors (TBR), using a new promising catalyst based on Pd/K2621. Excellent results were obtained from the experimental point of view, achieving high H2O2 selectivities, around 90% for a short contact time in the batch reactor, 60% in the semibatch reactor and 70% in the TBR. The simplest rate equations compatible with the acknowledged reaction network have been included in a reactor model, which accounts for mass transfer resistances between the gas and the liquid in the liquid–catalyst surface. The corresponding Arrhenius parameters were estimated from direct synthesis experiments for all the reactions and reactors. The models show how the reaction rates change among the batch, semibatch and trickle bed (TBR) reactors. The results suggest how to improve the reactor set-up and reaction performance in continuous operations and how to compare the results between the different reactors and conditions. The sensitivity analysis on the reaction allowed to gain new insights into the reaction rates. The TBR showed how the mass transfer limitations can help to direct the reaction towards the H2O2 synthesis. Remarkably, these results were achieved in the absence of any acids or halide ions, i.e. no known selectivity promoters for direct H2O2 synthesis were applied, thus the kinetics are not affected by the presence of promoters.


 Please wait while we load your content...
Please wait while we load your content...