Ultrafine Co3O4 nanolayer-shelled CoWP nanowire array: a bifunctional electrocatalyst for overall water splitting†
Abstract
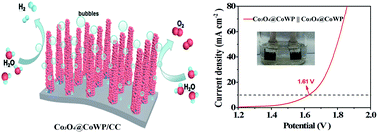
The development of bifunctional electrocatalysts based on highly efficient non-noble metals is pivotal for overall water splitting. Here, a composite electrode of Co3O4@CoWP is synthesized, where an ultrathin layer composed of Co3O4 nanoparticles is grown on CoWP nanowires supported on a carbon cloth (CC). The Co3O4@CoWP/CC electrode exhibits excellent electrocatalytic activity and improved kinetics towards both the oxygen and hydrogen evolution reactions (OER and HER). The Co3O4@CoWP/CC electrode achieves a current density of 10 mA cm−2 at a low overpotential of 269 mV for the OER and −10 mA cm−2 at 118 mV for the HER in 1.0 M KOH solution. The voltage applied to a two-electrode water electrolyzer for overall water splitting, while employing the Co3O4@CoWP/CC electrode as both an anode and a cathode, in order to reach a current density of 10 mA cm−2, is 1.61 V, which is better than that for the majority of reported non-noble electrocatalysts. Moreover, the Co3O4@CoWP/CC electrode exhibits good stability over 24 h with slight attenuation. The electrode benefits from the enhanced adsorption of oxygen intermediates on Co3O4 during the OER, the increased ability for water dissociation and the optimized H adsorption/desorption ability of CoWP nanowires during the HER. This study provides a feasible approach for cost-effective and high-performance non-noble metal bifunctional catalysts for overall water electrolysis.


 Please wait while we load your content...
Please wait while we load your content...