Preparation of cobalt sulfide@reduced graphene oxide nanocomposites with outstanding electrochemical behavior for lithium-ion batteries†
Abstract
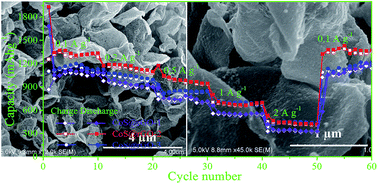
Cobalt sulfide@reduced graphene oxide composites were prepared through a simple solvothermal method. The cobalt sulfide@reduced graphene oxide composites are composed of cobalt sulfide nanoparticles uniformly attached on both sides of reduced graphene oxide. Some favorable electrochemical performances in specific capacity, cycling performance, and rate capability are achieved using the porous nanocomposites as an anode for lithium-ion batteries. In a half-cell, it exhibits a high specific capacity of 1253.9 mA h g−1 at 500 mA g−1 after 100 cycles. A full cell consists of the cobalt sulfide@reduced graphene oxide nanocomposite anode and a commercial LiCoO2 cathode, and is able to hold a high capacity of 574.7 mA h g−1 at 200 mA g−1 after 200 cycles. The reduced graphene oxide plays a key role in enhancing the electrical conductivity of the electrode materials; and it effectively prevents the cobalt sulfide nanoparticles from dropping off the electrode and buffers the volume variation during the discharge–charge process. The cobalt sulfide@reduced graphene oxide nanocomposites present great potential to be a promising anode material for lithium-ion batteries.


 Please wait while we load your content...
Please wait while we load your content...