A coumarin–dihydroperimidine dye as a fluorescent chemosensor for hypochlorite in 99% water†
Abstract
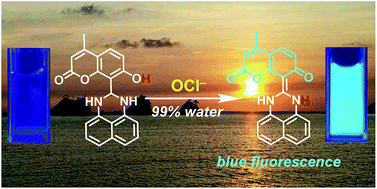
The hypochlorite anion (OCl−), a reactive oxygen species (ROS), is an important microbicidal agent in the immune system. Accurate and selective detection of OCl− in environmental and biological samples by a fluorescent molecular sensor is an important subject. All previously reported sensors, however, have suffered from tedious multi-step synthesis for the sensors and the use of large amounts of organic solvents for the analysis. Herein, we report that a coumarin–dihydroperimidine dye prepared by facile condensation behaves as a fluorescent sensor for OCl− in 99% water. The sensor exhibits weak fluorescence, but OCl−-selective dehydrogenation of its dihydroperimidine unit creates a strong blue fluorescence. This turn-on fluorescence response facilitates selective and sensitive detection of OCl− in the physiological pH range. Ab initio calculation revealed that the fluorescence enhancement by OCl− is triggered by intramolecular proton transfer from the coumarin –OH to the imine nitrogen of the formed perimidine moiety.


 Please wait while we load your content...
Please wait while we load your content...