Thermal stability of PMMA–LDH nanocomposites: decoupling the physical barrier, radical trapping, and charring contributions using XAS/WAXS/Raman time-resolved experiments†
Abstract
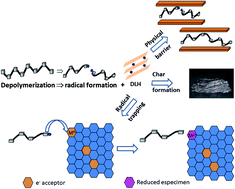
In-depth understanding of the thermal stability of polymer–clay nanocomposites requires the use of advanced time-resolved techniques combined with multivariate data analysis, as well as the preparation of layered nanofillers with well-defined composition. The layered double hydroxide (LDH) compounds Zn2Al(OH)6·nH2O, Zn2Al0.75Fe0.25(OH)6·nH2O, ZnCuAl(OH)6·nH2O, and ZnCuAl0.5Fe0.5(OH)6·nH2O were prepared, each designed to specifically identify the physical barrier, radical trapping, and char formation contributions to the thermal stability of the PMMA–LDH nanocomposites. The unique combination of conventional methods (TG, DSC, and Raman spectroscopy) and synchrotron radiation techniques (XAS and WAXS), applied during PMMA–LDH heating, revealed the synergetic (of iron) and antagonist (of copper) effects of the LDH layers transformations on the three main endothermic steps of mass loss of the polymer. The diffusion barrier effect was proved by the downshift of the PMMA thermal decomposition temperature caused by the decrease of the LDH edifice thermostability when divalent cations were substituted in the LDH (passing from PMMA–Zn2Al(OH)6·nH2O to PMMA–ZnCuAl(OH)6·nH2O). For PMMA–Zn2Al0.75Fe0.25(OH)6·nH2O, a cooperative contribution of iron reduction, stabilisation of layered edifice, and radical trapping effects was observed for the thermal stability of the nanocomposite. LDH also acted as a diffusion barrier to the efflux and evaporation of depolymerized species, favouring the charring which exerts an additional contribution to thermal stability of the PMMA–LDH nanocomposites.


 Please wait while we load your content...
Please wait while we load your content...