Synthesis and photocatalytic CO2 reduction performance of aminated coal-based carbon nanoparticles
Abstract
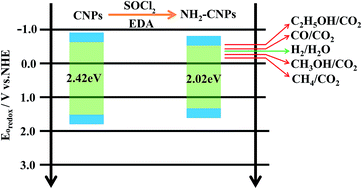
To obtain high-efficiency, low-cost, environmentally friendly carbon-based photocatalytic material, we synthesized coal-based carbon dots with sp2 carbon structure and multilayer graphene lattice structure by the hydrogen peroxide (H2O2) oxidation method to strip nano-scale crystalline carbon in the coal structure and link with oxygen-containing groups such as the hydroxyl group. N, S co-doped aminated coal-based carbon nanoparticles (NH2-CNPs) were then obtained by thionyl chloride chlorination and ethylenediamine passivation. The physical properties and chemical structure of the synthesized NH2-CNPs were studied and the photocatalytic CO2 reduction performance was tested. The results show that NH2-CNPs are vesicle-type spherical particles with particle size of 42.16 ± 7.5 nm and have a mesoporous structure that is capable of adsorbing CO2. A defect structure was formed on the surface of the NH2-CNPs due to the doping of N and S elements, thereby significantly improving the ability to photogenerate electrons under visible light along with the ability to efficiently separate the photo-generated carriers. The photocatalytic reduction products of CO2 over NH2-CNPs were CH3OH, CO, C2H5OH, H2 and CH4. After 10 hours of reaction, the total amount of products was 807.56 μmol g−1 cat, the amount of CH3OH was 618.7 μmol g−1 cat, and the calculated selectivity for conversion of CO2 to CH3OH was up to 76.6%.
- This article is part of the themed collection: Editors' collection: Carbon Dioxide Capture/Reduction


 Please wait while we load your content...
Please wait while we load your content...