Structural modification of oridonin via DAST induced rearrangement†
Abstract
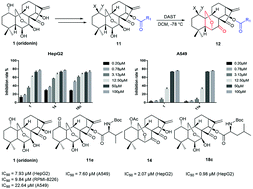
A simple and efficient protocol was developed for the syntheses of oridonin analogues, i.e. 6,20-epoxy ent-kaurane diterpenoid analogues from oridonin via diethylaminosulfur trifluoride (DAST) promoted rearrangement, most of which exhibited superior anticancer activities compared with their precursor.


 Please wait while we load your content...
Please wait while we load your content...