A reactive electrochemical filter system with an excellent penetration flux porous Ti/SnO2–Sb filter for efficient contaminant removal from water†
Abstract
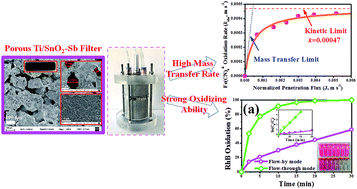
Tubular porous Ti/SnO2–Sb filters with excellent penetration flux (∼61.94 m3 m−2 h−1 bar−1) and electrochemical activity were prepared by a sol–gel method using low-cost porous titanium filters as the substrates. The porous Ti/SnO2–Sb filters were used as anodic reactive electrochemical membranes to develop reactive electrochemical filter systems, by combining membrane filtration technology with the electrooxidation process, for water treatment. A convection-enhanced rate constant of 4.35 × 10−4 m s−1 was achieved for Fe(CN)64− oxidation, which approached the kinetic limit and is the highest reported in an electrochemical system. The electrooxidative performance of the reactive electrochemical filter system was evaluated with 50 mg L−1 rhodamine B (RhB). The results showed that the reactive electrochemical filter system in flow-through mode resulted in an 8.6-fold enhancement in RhB oxidation as compared to those in flow-by mode under the same experimental conditions. A normalized rate constant of 5.76 × 10−4 m s−1 for RhB oxidation was observed at an anode potential of 3.04 V vs. SCE, which is much higher than that observed in a reactive electrochemical filter system with carbon nanotubes and/or Ti4O7 (1.7 × 10−5–1.4 × 10−4 m s−1). The electrical energy per order degradation (EE/O) for RhB was as low as 0.28 kW h m−3 in flow-through mode, with a relatively short residence time of 9.8 min. The overall mineralization current efficiency (MCE) was calculated to be 83.6% with ∼99% RhB removal and ∼51% TOC removal. These results illustrate that this reactive electrochemical filter system is expected to be a promising method for water treatment.


 Please wait while we load your content...
Please wait while we load your content...