Modeling alveolar injury using microfluidic co-cultures for monitoring bleomycin-induced epithelial/fibroblastic cross-talk disorder†
Abstract
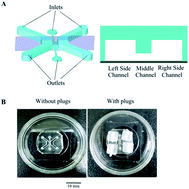
Epithelial/fibroblastic cross-talk is a significant disorder leading to pulmonary fibrosis, but its pathogenesis remains unclear because the current in vitro and in vivo models do not allow for optimal visualization of the complex disease processes at the human lung epithelial–interstitial interface. In this study we aimed to develop a microfluidic co-culture device that mimics a human disease model of pulmonary fibrosis. Our microfluidic device consists of two parallel side channels separated by a middle channel, recapitulating the physiological microenvironment of cell growth and the native microenvironment of the epithelial–interstitial interface in the human lung. We designed this device to represent bleomycin (BLM)-induced pulmonary fibrosis in human lungs. We recorded fibroblast and inflammatory cell migration in real-time and showed that epithelial cells became apoptotic or developed morphological changes in response to BLM. In summary, we established a co-culture device that models the native microenvironment of the lung epithelial–interstitial interface. Our data provide evidence of the role of epithelial cells and fibroblasts in pulmonary fibrosis, suggesting that fibrosis occurs as a result of injury to epithelial cells. Thus, epithelial injury plays an essential role in the pathogenesis of epithelial/fibroblastic cross-talk disorder.


 Please wait while we load your content...
Please wait while we load your content...