Shedding light on the effective fluorophore structure of high fluorescence quantum yield carbon nanodots†
Abstract
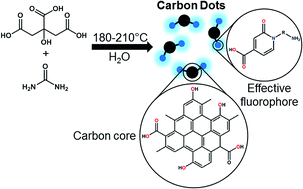
Carbon nanodots (CD) have great potential for imaging and sensing applications, due to their unique luminescence properties in combination with their low toxicity. Although CDs are currently the subject of intensive research activity, the exact structure of their effective fluorophore species has to be clarified because it can depend on the starting materials and reaction conditions. Most importantly, the limited photostability of some CDs presents an impediment to the much-needed breakthroughs required in the field. In this work, the photobleaching of CDs synthesized hydrothermally from citric acid and urea under various conditions is studied in detail. By visualizing changes in the absorption and fluorescence properties of CDs over time upon exposure to UV-light by means of in situ spectroscopic probes, we demonstrate a trade-off between high initial quantum yield and photostability. We found that the CDs consist of low-molecular weight fluorophores bound to π-conjugated domains (carbon core). In the case of CDs synthesized at higher reaction temperatures and/or for longer reaction times, lower initial quantum yields and higher photostability originate from partial conversion of the low-molecular weight fluorophores to π-conjugated domains during synthesis. Thus, the remaining surface bound fluorophores are bound to larger π-conjugated domains, which, in turn, enable dissipation of absorbed UV-energy and protection against photochemical damage. Reference experiments were performed with citrazinic acid – a pyridone-based structure – using ultrafast time-resolved spectroscopy, steady-state spectroscopy, and theoretical DFT simulations. We conclude that pyridone-like structures are most likely responsible for both the high quantum yields and the photobleaching of CDs.


 Please wait while we load your content...
Please wait while we load your content...