Solubility limits in quaternary SnTe-based alloys†
Abstract
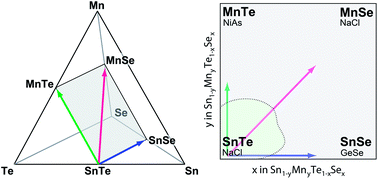
The controlled decomposition of metastable alloys is an attractive route to form nanostructured thermoelectric materials with reduced thermal conductivity. The ternary SnTe–MnTe and SnTe–SnSe heterostructural alloys have been demonstrated as promising materials for thermoelectric applications. In this work, the quaternary Sn1−yMnyTe1−xSex phase space serves as a relevant model system to explore how a combination of computational and combinatorial-growth methods can be used to study equilibrium and non-equilibrium solubility limits. Results from first principle calculations indicate low equilibrium solubility for x,y < 0.05 that are in good agreement with results obtained from bulk equilibrium synthesis experiments and predict significantly higher spinodal limits. An experimental screening using sputtered combinatorial thin film sample libraries showed a remarkable increase in non-equilibrium solubility for x,y > 0.2. These theoretical and experimental results were used to guide the bulk synthesis of metastable alloys. The ability to reproduce the non-equilibrium solubility levels in bulk materials indicates that such theoretical calculations and combinatorial growth can inform bulk synthetic routes. Further, the large difference between equilibrium and non-equilibrium solubility limits in Sn1−yMnyTe1−xSex indicates these metastable alloys are attractive in terms of nano-precipitate formation for potential thermoelectric applications.


 Please wait while we load your content...
Please wait while we load your content...