Decoloration of azo and triarylmethane dyes in the aqueous phase by catalytic hydrotreatment with Pd supported on pillared clays
Abstract
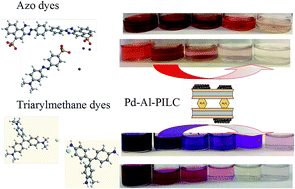
Catalytic hydrotreatment of synthetic dyes as azo compounds and triarylmethanes has been carried out under ambient-like conditions (25–50 °C, 1 atm) with H2 using Pd supported on Al-pillared clays and Al2O3 as catalysts. The treatment leads to complete decoloration of aqueous solutions of the azo dyes (azobenzene, methyl orange, Congo red and oil red) due to the catalytic hydrogenation of the N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bonds. Under acid pH the color removal was enhanced and a solid residue was formed in the case of Congo red, being completely removed. With aqueous solutions of the triarylmethane dyes crystal violet, fuchsine and malachite green complete decoloration was also achieved upon hydrogenation of the chromophore groups or modification of the chemical structure.
N bonds. Under acid pH the color removal was enhanced and a solid residue was formed in the case of Congo red, being completely removed. With aqueous solutions of the triarylmethane dyes crystal violet, fuchsine and malachite green complete decoloration was also achieved upon hydrogenation of the chromophore groups or modification of the chemical structure.


 Please wait while we load your content...
Please wait while we load your content...