Preparation of magnetic metal organic framework composites for the extraction of neonicotinoid insecticides from environmental water samples
Abstract
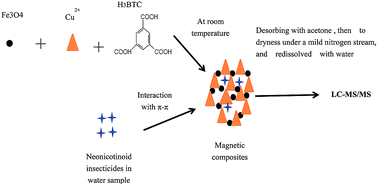
MOF-199/Fe3O4 nanoparticles were explored for the first time to purify environmental water samples and adsorb neonicotinoid insecticides as hybrid adsorbents. The composites were successfully synthesized by an in situ method at room temperature with electrostatic interaction to chemically stabilize the nanoparticles and metal ions. The nanoparticles were uniformly encoded with metal organic frameworks (MOFs). With the lowest loading amount of Fe3O4 applied to magnetic solid-phase extraction (MSPE) of six neonicotinoid insecticides in environmental water samples followed by high performance liquid chromatography tandem mass spectrometry (HPLC-MS/MS) analysis, the extraction efficiency was the highest. The main influencing factors including the solution ionic strength, solution pH, extraction time, desorbing organic solvent, and solvent volume, were also evaluated. Considering the adsorbing conditions and insecticide structures, the adsorbing mechanism was preliminarily discovered to be largely dependent upon the π–π interaction with the benzene ring in MOF-199 and delocalized large π bonds in the insecticide molecules. Under optimal conditions, the limits of detection (LODs) were 0.3–1.5 ng mL−1 with a signal-to-noise ratio (S/N) of 3. All the analytes exhibited good linearity with correlation coefficients (r2) of higher than 0.9947. The relative standard deviations (RSDs) for six neonicotinoid insecticides in environmental samples in five replicates ranged from 1.5% to 11.6%, and good recoveries from 88.0% to 107.0% were obtained, indicating that the MOF-199/Fe3O4 composites are feasible for analysis of trace analytes in environmental water samples.

 Please wait while we load your content...
Please wait while we load your content...