Molecularly imprinted solid phase extraction in an efficient analytical protocol for indole-3-methanol determination in artificial gastric juice†
Abstract
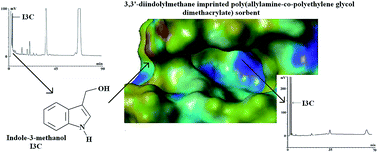
In this study we presented a new and efficient analytical protocol for determination of indole-3-methanol (indole-3-carbinol, I3C) in spiked artificial gastric juice used as a model sample. The analytical protocol involves separation of I3C on the imprinted stationary phase as well as the analysis of I3C using high performance liquid chromatography coupled with UV detection. The described analytical protocol allows selective isolation of I3C present in a complex matrix (such as inorganic salts and protein) from the mixture of I3C metabolites formed in artificial gastric juice with high total recovery equal to 96 ± 3% in a low concentration range of 0.125–10 μmol L−1. The limit of detection and limit of quantification were 0.150 and 0.454 μmol L−1, respectively. The efficacy of the analytical protocol was demonstrated by the results obtained for non-imprinted commercial sorbents C18, Florisil, and MCX Oasis® used in the separation step. The affinity of the imprinted polymer matrix was tested by non-competitive binding experiments of four structurally related analytes. The template behavior was analyzed during the imprinting process.

 Please wait while we load your content...
Please wait while we load your content...