Manganese silicate drapes as a novel electrode material for supercapacitors†
Abstract
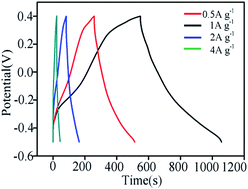
Manganese silicate was fabricated via a facile hydrothermal reaction between Na2SiO3·9H2O and MnCl2·4H2O. The morphology and microstructure of the as-prepared products were characterized by X-ray diffraction, transmission electron microscopy, Fourier transform infrared spectroscopy, X-ray photoelectron spectroscopy and nitrogen adsorption/desorption isotherms. The electrochemical properties of the manganese silicate were evaluated by cyclic voltammetry, galvanostatic charge/discharge and electrochemical impedance spectroscopy. With increasing the hydrothermal time from 0.5 to 3 h, the morphology of the product converted from aggregates composed of nanoparticles into thin drapes with an amorphous and porous structure. After hydrothermal reaction for 3 h, the porous product with a large specific surface area of 439 m2 g−1 exhibits a specific capacitance of 281 F g−1 at a current density of 1 A g−1 in 1 M KOH electrolyte as an electrode material for supercapacitors (SCs). Meanwhile, the product also demonstrates excellent rate performance. Thus the manganese silicate will be an alternative candidate for the electrode material of SCs.

 Please wait while we load your content...
Please wait while we load your content...