MOF-derived Fe3O4/carbon octahedral nanostructures with enhanced performance as anode materials for lithium-ion batteries
Abstract
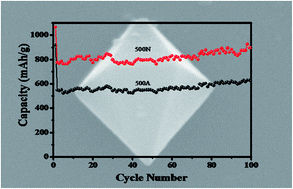
Fe3O4/carbon (C) nanocomposites with octahedral structure have been successfully prepared as anode materials for lithium-ion batteries (LIBs) by carbonizing hexagonal dipyramid Fe–metal organic frameworks (MOFs) under N2 directly. The prepared Fe–MOFs and Fe3O4/C octahedral nanostructures were carefully characterized by scanning electron microscopy, energy dispersive X-ray spectroscopy, X-ray powder diffraction, thermogravimetric analysis, N2 adsorption/desorption isotherms and electrochemical techniques. The results showed that octahedral Fe3O4 was encapsulated by carbon layers to form porous Fe3O4/C octahedral nanostructures. Benefiting from the high electrical conductivity of the carbon matrix and the porous structure of Fe3O4/C nanocomposites, the Fe3O4/C octahedral nanostructures demonstrated a lithium storage capacity of 861 mA h g−1 after 100 cycles at a specific current of 100 mA g−1, as anode for lithium ion batteries (LIBs), which is much higher than that of spindle-like Fe2O3 obtained via direct carbonization of Fe–MOFs under an air atmosphere (merely 603 mA h g−1 under identical conditions) and commercial Fe3O4. The elaborate and well-defined configuration also delivered a high rate capability and long cycling stability. Hence, the combination of Fe3O4 crystals with an octahedral nanostructure and the random doping of carbon could offer a valid method for the development of anode materials for high-performance LIBs.

 Please wait while we load your content...
Please wait while we load your content...