Magnesium cofactor produces unpaired electrons confined by triplet nucleotide loops in a full-turn DNA fragment
Abstract
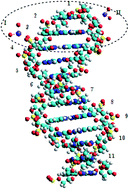
The B-DNA curvature arising from pairing between nucleotides of the two curved complementary DNA strands affects the oxidation number of magnesium cofactor. When the reading frame (RF) spans over three paired nucleotides (5′-3′ and 3′-5′ strands), the magnesium cations on both strands become singly charged. This produces two radical pairs (RPs) with four electrons – two electrons on Mg+ and two electrons on two complementary nucleotide triplets. Thanks to curvature and hyperfine coupling (interaction between the active nuclear spin on the 31P atom of each nucleotide and the RP electrons), the four electrons of the two RPs lose their initial energy equality. The two electrons (the first RP) collapse forming the low energy inactive singlet state while the other two electrons (the second RP) remain uncoupled. These two electrons slowly rotate over the triplet nucleotide frame in opposite directions (the Rashba effect) forming two conduction loops. The shift of the RF to the next three complementary nucleotides results in reversing the rotations. The reverse of rotation makes the loops stand separately over the full length of DNA helix. Each RF shows its unique direction of spin polarization, which being mapped onto the unit vector, depending on the curvature, produces a topological phase/sector. Each phase is directly linked to one of the twenty canonical amino acids. The results rest on QM/MM computations where the QM part is treated with the DFTB3LYP method in curvilinear coordinates. The finding has practical implementation: constructing non-dissipative nanochips for quantum computing.

 Please wait while we load your content...
Please wait while we load your content...