Photoelectrochromic devices based on cobalt complex electrolytes
Abstract
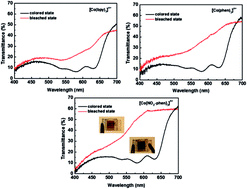
In this paper, a photoelectrochromic device (PECD) composed of a conducting polymer electrochromic electrode and a ruthenium-based dye-sensitized TiO2 nanoparticle electrode is constructed and investigated. Three cobalt complexes, which are [Co(bpy)3]n+, [Co(phen)3]n+ and [Co(NO2-phen)3]n+, are introduced into the PECD as the redox couples and a self-powered coloring/bleaching process of the cobalt electrolyte-based PECDs is achieved for the first time. The PECD using the [Co(NO2-phen)3]n+ electrolyte presents the highest transmittance modulation of 42.6% at 636 nm between the colored and bleached states. From current–voltage characteristics, it is found that the transmittance of bleached PECD is improved with a higher open-circuit voltage, resulting in an enhanced transmittance modulation. Electrochemical impedance spectroscopy (EIS) studies indicate that the charge transfer resistance (Rct) at PProDOT-Me2|electrolyte interface is correlated with the response time for the bleaching process.

 Please wait while we load your content...
Please wait while we load your content...