An in situ self-catalytic hybrid cyanate ester resin and its self-catalytic polymerization behavior†
Abstract
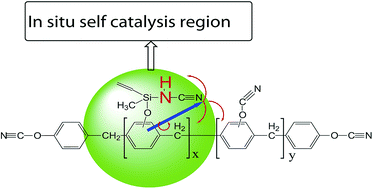
A novolac cyanate ester (NCE) with self-catalytic function that incorporates –Si–NH–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N group has been synthesized through the reaction between methylvinylcyclotrisilazane (MVSZ) and novolac cyanate ester (NCE). The analysis of polymerization by in situ Fourier transform-infrared spectroscopy (FT-IR) reveal that the catalytic polymerization is dominated by the cyclotrimerization of –OCN and trimerization between NH–C
N group has been synthesized through the reaction between methylvinylcyclotrisilazane (MVSZ) and novolac cyanate ester (NCE). The analysis of polymerization by in situ Fourier transform-infrared spectroscopy (FT-IR) reveal that the catalytic polymerization is dominated by the cyclotrimerization of –OCN and trimerization between NH–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N and –OCN, and the asymmetric and symmetric triazine ring were formed. The investigation of self-catalytic performance of O–Si–NH by differential scanning calorimetry (DSC) indicate that the O–Si–NH could significantly decrease the polymerization temperature, catalyze the polymerized reaction not only at lower temperature but also higher temperature even the limit from network structures, and endow the polymerized hybrid NCE with higher polymerized degree. Through dynamic mechanical analysis (DMA) analyses, it is concluded that the catalytic effect of –O–Si–NH– could improve the storage modulus and reduce the internal stress produced from polymerization process. The introduction of –Si–NH–C
N and –OCN, and the asymmetric and symmetric triazine ring were formed. The investigation of self-catalytic performance of O–Si–NH by differential scanning calorimetry (DSC) indicate that the O–Si–NH could significantly decrease the polymerization temperature, catalyze the polymerized reaction not only at lower temperature but also higher temperature even the limit from network structures, and endow the polymerized hybrid NCE with higher polymerized degree. Through dynamic mechanical analysis (DMA) analyses, it is concluded that the catalytic effect of –O–Si–NH– could improve the storage modulus and reduce the internal stress produced from polymerization process. The introduction of –Si–NH–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N to produce the catalytic effect for cyanate ester system is a promising method for modifying disadvantages of cyanate ester during polymerization.
N to produce the catalytic effect for cyanate ester system is a promising method for modifying disadvantages of cyanate ester during polymerization.

 Please wait while we load your content...
Please wait while we load your content...