Microwave-assisted synthesis of BiFeO3 nanoparticles with high catalytic performance in microwave-enhanced Fenton-like process†
Abstract
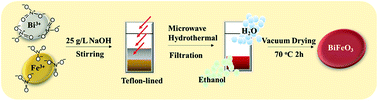
In this study, BiFeO3 (BFO) was successfully prepared by microwave-assisted hydrothermal synthesis (MAHS) and applied for the degradation of rhodamine B (RhB). The optimum experiment conditions for synthesizing BFOs using MAHS were obtained with the highest removal rate and degradation rate constant of RhB. Moreover, the synthesis time could be largely shortened through adopting microwave irradiation (MW). And then, in order to prove the superiority on BFO-MAHS, traditional hydrothermal method (THM) was used to compare with MAHS including the characterization analysis, the performances of produced hydroxyl radical (OH·) and the other advantages. The results reveal that BFO-MAHS has higher crystallinity, specific surface area and catalytic activity than BFO-THM due to a specific radiation effect and non-thermal effect of microwave during the BFO synthesis. These findings indicated that the feasible preparation of BFO-MAHS offers a new insight into the synthesis of high catalytic activity BFO which was applied in wastewater treatment.

 Please wait while we load your content...
Please wait while we load your content...