Magnetically separable CuFe2O4/reduced graphene oxide nanocomposites: as a highly active catalyst for solvent free oxidative coupling of amines to imines†
Abstract
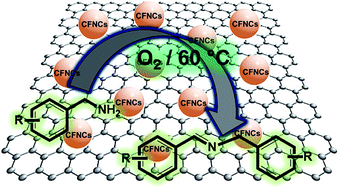
In this report, a very simple and straightforward one step coprecipitation method was used for decorating CuFe2O4 nanoparticles (NPs) over graphene sheets. The as synthesized CuFe2O4/RGO nanocomposites (CFRNCs) were fully characterized by XRD, TEM, HRTEM, XPS, FTIR, Raman, ICP-AES and SEM-EDS analysis which shows well controlled small sized (∼7 nm from Scherrer formula) CuFe2O4 NPs with dense and compact loading over RGO sheets. The magnetic study reveals the superparamagnetic behavior of ferrite NPs which provides an extra benefit in catalytic recyclability. The synthesized CFRNCs were found to be highly active in aerobic oxidative coupling of amines to desired imines achieving excellent yields (88–95%) under mild reaction conditions. Moreover the role of Cu, Fe species and their interaction with RGO sheets was well studied separately by synthesizing CuO/RGO, Fe2O3/RGO, bare CuFe2O4 and RGO samples, then catalytic mechanism was proposed based upon various controlled reaction results. The CFRNCs were recycled up to 4th cycle under similar catalytic conditions and found to be highly efficient without any accountable loss in activity and stability. This outstanding activity of CFRNCs can be attributed to synergistic coupling between Cu/Fe species, high surface area due to the presence of RGO sheets and small sized surfactant free CuFe2O4 NPs which led to the use of the present nanocatalyst in many more industrially important catalytic applications.

 Please wait while we load your content...
Please wait while we load your content...