Atomically precise and monolayer protected iridium clusters in solution†
Abstract
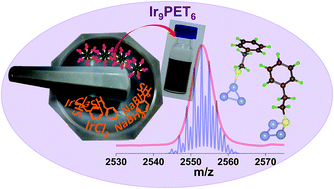
The first atomically precise and monolayer protected iridium cluster in solution, Ir9(PET)6 (PET – 2-phenyethanethiol) was synthesized via a solid state method. The absence of a plasmonic band at ∼350 nm, expected in the UV/Vis spectra for spherical Ir particles of 10 nm size indicated that the synthesized cluster is smaller than this dimension. Small angle X-ray scattering (SAXS) showed that the cluster has a particle size of ∼2 nm in solution which was confirmed by transmission electron microscopy (TEM). The blue emission of the cluster is much weaker than many noble metal clusters investigated so far. X-ray photoelectron spectroscopy (XPS) measurements showed that all Ir atoms of the cluster are close to the zero oxidation state. The characteristic S–H vibrational peak of PET at 2560 cm−1 was absent in the FT-IR spectrum of the cluster indicating RS–Ir bond formation. The molecular formula of the cluster, Ir9(PET)6 was assigned based on the most significant peak at m/z 2553 in the matrix assisted laser desorption ionization mass spectrum (MALDI MS), measured at the threshold laser intensity. Density functional theory calculations on small Ir@SCH3 and Ir@PET clusters and comparison of the predictions with the IR and 1H-NMR spectra of Ir9(PET)6 suggested that the PET ligands have two distinct structural arrangements and are likely to be present as bridging thiolates –(Ir–SR–Ir)– and singly attached thiolates –(Ir–SR).

 Please wait while we load your content...
Please wait while we load your content...