Optimization of the process parameters for the adsorption of ternary dyes by Ni doped FeO(OH)-NWs–AC using response surface methodology and an artificial neural network†
Abstract
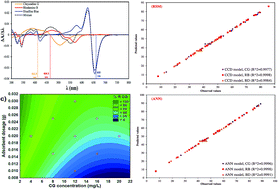
The present study deals with the simultaneous removal of chrysoidine G (CG), rhodamine B (RB) and disulfine blue (DB) by Ni doped ferric oxyhydroxide FeO(OH) nanowires on activated carbon (Ni doped FeO(OH)-NWs–AC). The adsorbent was characterized using X-ray diffraction (XRD), field emission scanning electron microscopy (FE-SEM) and scanning electron microscopy (SEM). Derivative spectrophotometry was used for investigation of simultaneous dye adsorption by an artificial neural network (ANN) and response surface methodology (RSM) to analyse and model their adsorption behavior. Using the ANN analysis, the optimal configuration of the ANN model for modeling of the adsorption process was found to be (6:(4–6):3). The effect of adsorption parameters such as initial pH, adsorbent mass, sonication time and initial CG, RB and DB concentration was studied using central composite design (CCD), while design results were also utilized as a training set for the ANN. After predicting the model using RSM and ANN, the two methodologies were statistically compared by their coefficient of determination, root mean square error, absolute average deviation and mean absolute error based on the validation data set. Results suggest that ANN has better prediction performance as compared to RSM. It was also found that response surface methodology (RSM) predicts the suitability of output parameters. The adsorption mechanism and process rates were investigated by analyzing time dependency data using various conventional kinetic models such as pseudo-first-order and second order, intra-particle diffusion and Elovich models and the best fit was obtained by a pseudo-second-order kinetic model with good agreement between the equilibrium and expected adsorption data. The experimental results revealed that dye adsorption was highly linear and followed the Langmuir isotherm model with maximum adsorption capacities of 187.420 (CG), 210.170 (RB) and 235.650 mg g−1 (DB).
- This article is part of the themed collections: Machine learning and artificial neural networks in chemistry and Adsorption and degradation of pollutants

 Please wait while we load your content...
Please wait while we load your content...