Photodegradation of organic dyes based on anatase and rutile TiO2 nanoparticles†
Abstract
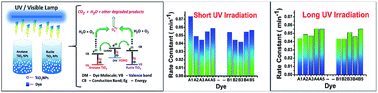
The present work highlights a simple nanochemistry based clean and efficient process for effective degradation of various organic dyes by use of anatase and rutile TiO2 nanoparticles. Based on photodegradation studies it was observed that at certain experimental parameters, rutile TiO2 was as effective as anatase for the degradation of indigo carmine (IC) dye in aq. solution. However, anatase TiO2 was found to be much more efficient against methylene blue (MB), methyl orange (MO), rhodamine B (RB) and eriochrome black T (EBT) under short UV irradiation. The photodegradation study of these organic dyes was also performed under long UV irradiation employing both anatase and rutile titania and the results showed similar trends. However, only EBT photodegradation indicated equal photocatalytic activity by both phases. The catalytic degradation of the organic dyes was studied by degradation kinetics. The maximum degradation efficiency using anatase and rutile titania photocatalysts was found to be 88% and 77% in the case of MB under short UV irradiation while about 65% was found in the case of EBT under long UV irradiation. The reuse of photocatalyst even after five cycles retained the degradation efficiency of 83% and 71% respectively for anatase and rutile when tested against MB.

 Please wait while we load your content...
Please wait while we load your content...