Unprecedented inhibition of glycosidase-catalyzed substrate hydrolysis by nanodiamond-grafted O-glycosides†
Abstract
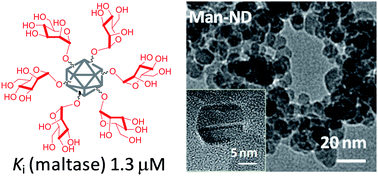
We report herein the unprecedented finding that α-O-glucosides and also α-O-mannosides, when conjugated on nanodiamond particles (ND), are not only stable towards the hydrolytic action of the corresponding matching glycosidases, but are also endowed with the ability to inhibit them. Moreover, conjugation of the O-glycosides to ND (glyco-ND) sees them transformed into inhibitors of mismatching enzymes (for which they do not serve as substrates even when in their monovalent, free form). The effects of the glyco-NDs have been demonstrated on a panel of commercial glycosidases and the inhibition found to be competitive and reversible and not to be related to any denaturation of enzymes by the ND-conjugates. Values for Ki in the low micromolar range have been measured for certain glyco-ND (for example, a Ki value of 5.5 ± 0.2 μM was measured for the glucopyranosyl-coated NDs against the α-glucosidase from baker's yeast) and found to depend on both the identity of the enzyme and the glyco-ND. The latter Ki value compares well with that obtained for the natural glucosidase inhibitor, 1-deoxynojirimycin (Ki of 25 μM against the α-glucosidase from baker's yeast under identical assay conditions). The monovalent control O-glycosides was hydrolysed efficiently by the appropriate glycosidase. Glyco-ND bearing 50% loading of O-glycoside as well ND conjugated with both O-glucosides and O-mannosides (mixed) have also been assayed and shown also to inhibit the panel of glycosidases with potencies and selectivities different from those recorded for the 100% loaded ND and also from one another. The impact on factors such as glycotope density and heteromultivalency on inhibition is reminiscent of that typically encountered in carbohydrate–lectin recognition events. The abilities of the glyco-ND to bind, cross-link and aggregate concanavalin A, a lectin known to recognize both α-O-D-mannosides and α-O-D-glucosides, was assessed by a range of methods including an enzyme-linked lectin assay (ELLA), a two-site sandwich ELLA and a turbidimetry assay, respectively and indeed seen to reflect their expected per glycotope affinity enhancements as compared to monovalent controls: the high avidity of the lectin for each respective glycosylated ND particle was consistent with the manifestation of potent multivalent effects driving lectin recognition and binding.

 Please wait while we load your content...
Please wait while we load your content...