Experimental and theoretical studies of triisopropanolamine as an inhibitor for aluminum alloy in 3% NaCl solution
Abstract
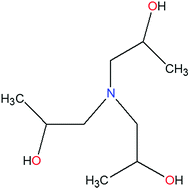
The inhibition performance and mechanism of triisopropanolamine (TIPA) for the corrosion of aluminum alloy in 3% NaCl solution were investigated using weight loss method, potentiodynamic polarization, electrochemical impedance spectroscopy (EIS) and scanning electron microscope (SEM). The electrochemical results revealed that TIPA was an effective inhibitor and the inhibition efficiency increased with the increasing concentrations of TIPA, which was further confirmed by SEM observation. Potentiodynamic polarization studies revealed that TIPA acted as a mixed-type inhibitor. The adsorption of TIPA on the aluminum surface obeyed a Langmuir thermodynamic–kinetic model, and the thermodynamic and kinetic parameters meant that the interaction between the inhibitor and the surface of aluminum alloy involved both physisorption and chemisorption. Moreover, theoretical calculations were used to investigate inhibition mechanism of the studied inhibitor. The results showed that TIPA molecule could adsorbed on Al substrate spontaneously and chelated with Al atom due to the hydroxyl group.

 Please wait while we load your content...
Please wait while we load your content...