Improved structural and electrochemical performances of LiNi0.5Mn1.5O4 cathode materials by Cr3+ and/or Ti4+ doping†
Abstract
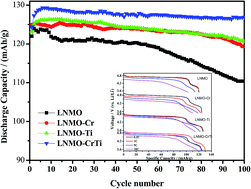
High voltage LiNi0.45M0.05Mn1.5O4 (M = Ni, Cr, Ti, Cr0.5Ti0.5) cathode materials were synthesized by solid-state method, and the effects of Cr/Ti doping alone and co-doping on the crystalline structure, Mn3+ content, particle morphology and electrochemical performance of LiNi0.5Mn1.5O4 cathode materials were systematically investigated. The as-prepared samples were characterized by XRD, FT-IR, SEM, CV, EIS and galvanostatic charge/discharge cycling tests. XRD results show that both pristine and doped materials have cubic spinel structure with Fd3m space group, and the Cr and/or Ti doping can effectively prevent the formation of LiyNi1−yO impurity phase. FT-IR spectra indicate that the Cr and/or Ti doping increases the disordering degree of Ni/Mn ions in 16d octahedral sites. SEM observation discloses that the Cr and/or Ti doping increases the particle size distribution homogeneity and decreases the average primary particle size. EIS analysis illustrates that the Cr and/or Ti doping decreases the charge transfer resistance and increases the Li+ ion diffusion coefficient. All of the above-mentioned factors are believed to be advantageous to the cycling stability and rate capability. Among which, the Cr and Ti co-doped sample LiNi0.45Cr0.025Ti0.025Mn1.5O4 exhibits optimal cycling performance with a capacity retention rate of 102.1% after 100 cycles at 1C rate, and optimal rate capability with a discharge capacity of 118.7 mA h g−1 at 10C rate, which is 96.1% of its capacity at 0.2C rate. The excellent electrochemical performance of LiNi0.45Cr0.025Ti0.025Mn1.5O4 cathode material may be mainly attributed to the presence of appropriate Mn3+ content and higher Li+ ion diffusion coefficient.

 Please wait while we load your content...
Please wait while we load your content...