Optimization of production conditions for synthesis of chemically activated carbon produced from pine cone using response surface methodology for CO2 adsorption
Abstract
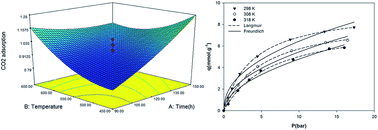
The conditions for the preparation of activated carbon from pine cone for CO2 adsorption were optimized by response surface methodology (RSM) and central composite design (CCD). The effects of the activation time, activation temperature, and impregnation ratio of H3PO4 were studied. The produced activated carbons were characterized by CO2 adsorption capacity, iodine number and carbon yield. Three quadratic models were developed to correlate the preparation variables for three responses. The experimental data obtained from activated carbon prepared under optimum condition were in good agreement with the values predicted from the models, with relatively small errors between the predicted and the actual values. The prepared activated carbon was characterized by elemental analysis, N2 adsorption isotherm at 77 K, Fourier transform infrared spectroscopy (FTIR), transmission electron microscopy (TEM) and scanning electron microscopy (SEM). The equilibrium adsorption of CO2 on prepared AC was experimentally investigated via volumetric method at temperature range of 298–318 K and pressures up to 16 bars. The experimental CO2 adsorption data were analyzed by Langmuir and Freundlich isotherms models. The adsorption behavior was well described by the Langmuir isotherm model, showing a monolayer adsorption capacity for CO2 on prepared AC. Results demonstrated that the negative values of heat of adsorption obtained for AC were lower than 80 kJ mol−1 indicating that the interaction between AC and CO2 molecules is exothermic and physical interactions.

 Please wait while we load your content...
Please wait while we load your content...