Dual energy transfer controlled photoluminescence evolution in Eu and Mn co-activated β-Ca2.7Sr0.3(PO4)2 phosphors for solid-state lighting†
Abstract
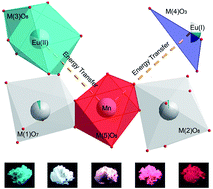
Photoluminescence evolution in single-composition phosphors for solid state lighting can be realized through an energy transfer strategy, which is commonly based on one sensitizer and one activator. In this work, we focus on a dual energy transfer process, that is, energy transfers from the sensitizer Eu2+ at two different cation sites to the activator Mn2+ at another site in whitlockite-type β-Ca2.7Sr0.3(PO4)2. Firstly, we refined the crystal structure of the β-Ca2.7Sr0.3(PO4)2 host by a Rietveld method. Then we studied the phase, morphology and photoluminescence of Eu and Mn co-activated β-Ca2.7Sr0.3(PO4)2 phosphors, which are synthesized by a conventional solid-state reaction. By adjusting the ratio of Eu2+/Mn2+, the emission hue can be controlled from cyan (0.211, 0.281) to white-light (0.332, 0.290) and eventually to red (0.543, 0.276). The dual energy transfers from Eu2+ to Mn2+ are demonstrated to be dipole–dipole and quadrupole–quadrupole mechanisms, respectively. Moreover, white light-emitting diodes (LEDs) were fabricated through the integration of a 375 nm NUV chip and the white-emitting phosphor Ca2.7Sr0.3(PO4)2:0.008Eu2+, 0.03Mn2+ into a single package, which shows a warm white light with color coordinates of (0.40, 0.41) and correlated color temperature of 3731 K.

 Please wait while we load your content...
Please wait while we load your content...