Role of acidic sites in beta-hexachlorocyclohexane (β-HCH) adsorption by activated carbons: molecular modelling and adsorption–desorption studies
Abstract
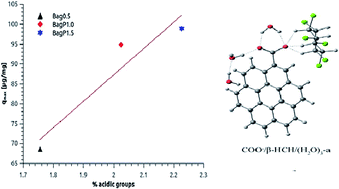
The removal of β-HCH from contaminated water by adsorption on activated carbons is presented. Sugar cane bagasse activated carbons with different textural and surface chemical properties were prepared and characterized for studying the β-HCH adsorption process. The isotherms are correlated by six models, among which the Fowler–Guggenheim/Jovanovic–Freundlich model is found to provide the best fit for two of the studied activated carbons. The isotherm adsorption data were correlated with the surface functional group composition. β-HCH adsorption was favored by the presence of acidic groups at the AC surface of chemically activated carbons. A thorough theoretical exploration of the potential energy surface for the interaction of β-HCH with a graphene sheet alone or containing hydroxyl or carboxyl surface groups was carried out. Molecular modelling results showed that under neutral to slightly acidic conditions only carboxylic surface groups should significantly contribute to β-HCH adsorption, in qualitative agreement with the experimental results. The analysis of the most stable structures suggests that the hydrogen bonds between the axial hydrogens of β-HCH and the charged oxygens of the deprotonated acidic surface groups are the main interactions responsible for the adsorption. This result is confirmed by a Temperature-Programmed Desorption study, showing that β-HCH molecules are associated with carboxylic groups of the AC surface.

 Please wait while we load your content...
Please wait while we load your content...