Ab initio investigations on the gas phase basicity and nonlinear optical properties of FLinOH species (n = 2–5)†
Abstract
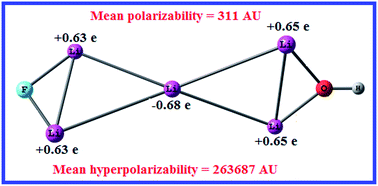
Superalkalies, due to lower ionization potentials than alkalies, possess potential reducing capability and can be used in the synthesis of a variety of charge transfer salts. In order to check whether superalkali hydroxides are as basic as a typical alkali hydroxide, we have performed a systematic investigation on the hydroxides of small FLin (n = 2–5) superalkali clusters. The equilibrium structures are identified and their stability is analyzed against the elimination of LiOH and LiF molecules. Our MP2 calculations show that the proton affinity and gas phase basicity of the FLinOH species are lower than that of LiOH by 50–100 kJ mol−1. The trend of basicity of FLinOH is in accordance with their HOMO–LUMO energy gap as well as the LiOH and LiF elimination energies. Considering the fact that charge transfer salts possess significant nonlinear optical (NLO) responses, we have also calculated NLO parameters such as the total static dipole moment, the mean polarizability and the first static mean hyperpolarizability of the FLinOH species. The dipole moment and the polarizability values increase successively from FLi2OH to FLi5OH. On the contrary, the hyperpolarizability only increases for FLi3OH (2.7 × 103 a.u) and FLi5OH (2.6 × 105 a.u.). The dramatically large hyperpolarizability of FLi5OH is due to anionic Li possessing an excess electron, thus resembling the feature of an “alkalide”. The present work should provide new insights into the design of strong inorganic bases for chemical synthesis as well as potential NLO species for electro-optical applications.


 Please wait while we load your content...
Please wait while we load your content...