Novel red blood cell shaped α-Fe2O3 microstructures and FeO(OH) nanorods as high capacity supercapacitors†
Abstract
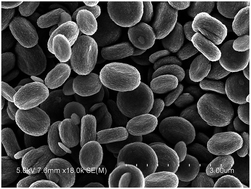
In this paper, we report the synthesis of novel red blood cell shaped α-Fe2O3 microstructures for the first time, which were synthesized by using urea as a hydrolysis agent, ammonium chloride as a structure directing agent and PVA as a surfactant through a hydrothermal method. By only adjusting the hydrothermal temperature, FeO(OH) nanorods were obtained. When used as electrode materials in supercapacitors, the FeO(OH) nanorods exhibit much better capacitive properties of 392.6 F g−1 than red blood cell shaped α-Fe2O3 microstructures (118.1 F g−1) at a current density of 1 A g−1, maintaining 91.4% and 75.5% of the initial capacity at a current density of 1 A g−1 after 500 cycles respectively. Such high performance can be attributed to the desirable morphology and components. The results manifest that the FeO(OH) nanorods may be promising electrode materials for supercapacitors.

 Please wait while we load your content...
Please wait while we load your content...