Exploring the mechanism of isomerisation and water-migration in the water-complexes of amino-acid l-proline: electrostatic potential and vibrational analysis†
Abstract
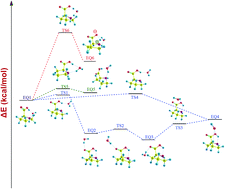
Chemical pathways for the gas-phase isomerisation between the four lowest-lying conformers of amino acid L-proline are proposed using the computations performed through the advanced quantum-mechanical methods like Hartree–Fock, density functional and coupled cluster theories. Besides this, the water-migration pathways in the complexes of L-proline with a single-water molecule are presented. The conformers and water-complexes of L-proline are characterized using spectral analysis involving vibrational frequencies, IR intensities and Raman scattering activities. The effect of electric charge distribution on the mechanism of isomerisation and water-migration is further analysed through the electrostatic potential. The detailed mechanism of the isomerisation in the water complexes, involving zwitterions, has also been unveiled which however is not feasible for the isolated L-proline. Notably, during the isomerisation process of the water-complexes, both positive and negative catalytic effect of the water is encountered. Interesting behaviour in the water-complexes is observed during the analysis of standard Gibbs free-energy change and its temperature-dependence. Besides this, the hydrogen-bonding interaction in the water-complexes of L-proline is also investigated through natural bond orbital analysis. The zwitterionic complexes of L-proline with single-water molecule have also been analysed through the Hirshfeld scheme as well as through widely used population analysis methods.


 Please wait while we load your content...
Please wait while we load your content...