Evaluation of undoped and M-doped TiO2, where M = Sn, Fe, Ni/Nb, Zr, V, and Mn, for lithium-ion battery applications prepared by the molten-salt method
Abstract
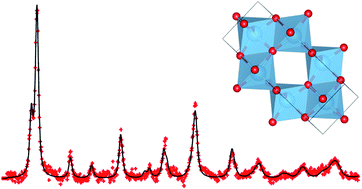
The molten-salt method was used to synthesize a series of transition-metal containing titanium dioxides. Some of the transition metals were found to substitute into the TiO2 lattice, such as (Ti0.9Fe0.1)O2, (Ti0.9Zr0.1)O2, (Ti0.9V0.1)O2, and (Ti0.9Mn0.1)O2, while others were formed as composite electrodes (in addition to relatively minor substitutions), namely 0.1SnO2–0.9TiO2 and 0.05NiO–0.1Nb2O5–0.9TiO2. Although identical synthesis-conditions were used the different transition metals yielded different phases. A comparative study of the electrodes relating surface area and composition (via X-ray photoelectron spectroscopy, XPS), and electrochemical behaviour is presented in this work. Among the substituted single phase electrodes, (Ti0.9Zr0.1)O2 exhibited the best reversible capacity of ∼160 mA h g−1, at the end of the 60th cycle in the voltage range 1.0–2.6 V, with a capacity fade of 24% from the 2nd to the 60th cycle. Among the composite electrodes, 0.05NiO–0.1Nb2O5–0.9TiO2 shows the best performance which is comparable to pure TiO2 but with a slower capacity-fade on extended cycling. The worst performing electrode is (Ti0.9V0.1)O2 with a reversible capacity of only ∼70 mA h g−1 at the end of 70 cycles with a current density of 130 mA g−1 in the voltage range 1.0–2.6 V and a capacity drop of 52% from the 2nd to the 70th cycle. The composite 0.1SnO2–0.9TiO2 features the highest irreversible capacity-loss. Zr-substitution into TiO2 gives the best electrochemical performance.

 Please wait while we load your content...
Please wait while we load your content...