Synthesis, chiroptical and SHG properties of polarizable push–pull dyes built on π-extended binaphthyls†
Abstract
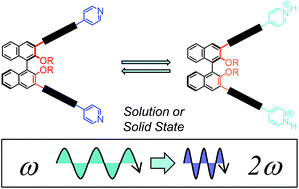
We report on new enantiopure binaphthyl derivatives in which electron-donating and electron-withdrawing substituents are placed in direct conjugation, to create active push–pull dyes for NLO applications. The dyes, unprecedentedly, extend their π-bridge from the 3,3′ positions of the binaphthyl units, and incorporate as acceptors pyridine units, possessing a coordinating nitrogen atom useful for further supramolecular polarization of the chiral dyes. The π-bridge is constructed by the sequential attachment of phenylenevinylene units to the enantiopure binaphthyl derivatives through Horner–Wadsworth–Emmons olefination, which proceed with high stereoselectivity, affording stereodefined chiral dyes. The polarization of the terminal pyridine units by means of labile complexation with Pd2+ ions has been demonstrated using both optical and chiroptical methods. The polarization by protonation can be made reversible in solution and solid state by exposure to ammonia vapors, as shown by absorption and emission spectroscopies. NLO properties, as determined by EFISH generation measurements in solution, are significant for the bisprotonated species when compared to previously reported binaphthyl substrates. TDDFT calculations show that the hyperpolarizability tensor contribution is responsible for enhancing SHG values upon protonation up to one order of magnitude, highlighting the potential of such ortho related, axially-chiral push–pull dyes for functional NLO applications.


 Please wait while we load your content...
Please wait while we load your content...