Interaction of bio-relevant thio-ether and thiols with dinuclear Pd(ii) complex: kinetics, mechanism, bioactivity in aqueous medium and molecular docking†
Abstract
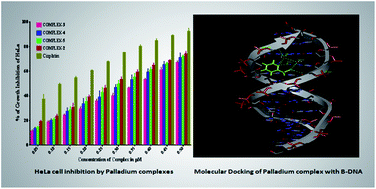
The kinetics of interaction between [Pd(pic)(OH)]2(ClO4)2, 2 (pic = 2-aminomethylpyridine) with the selected ligands (L) DL-methionine (DL-meth), L-cysteine (L-cys) and N-acetyl-L-cysteine (N-ac-L-cys) have been studied under pseudo-first order conditions using a stopped-flow spectrophotometer in aqueous medium as a function of [complex 2] as well as [ligand], pH, and temperature at constant ionic strength. The ligand dependent second order reaction is found to take place in two consecutive steps in accordance with the rate law, k(obs) = k1[L]2 in which all these three reactions follow the third order kinetics. The first step of the reaction is dependent, while the second step is independent of [ligand] in all the cases. The activation parameters, ΔH‡ and ΔS‡ for the two-step reactions are evaluated from the Eyring equation. An associative mode of activation (an associative mechanism) in the transition state is proposed for all these substitution processes. Complex 2 and its substituted products [Pd(pic)DL-meth]+ 3; [Pd(pic)L-cys] 4; and [Pd(pic)N-ac-L-cys] 5 are characterized by UV-Vis, FT-IR, 1H-NMR and ESI-mass spectroscopic methods. Complex 2–5 show remarkable anticancer properties on HeLa cells of about 70% at high concentration when compared to cis-platin and antibacterial properties on both the Gram positive (Bacillus subtilis) and Gram negative (E. coli Dh5α) bacteria. In addition, DNA interaction with plasmid DNA is observed and computational molecular docking studies were carried out with an aim to establish the binding mode of complex 2 with B-DNA.

 Please wait while we load your content...
Please wait while we load your content...