Towards an understanding of the role of hyper-branched oligomers coated on cathodes, in the safety mechanism of lithium-ion batteries†
Abstract
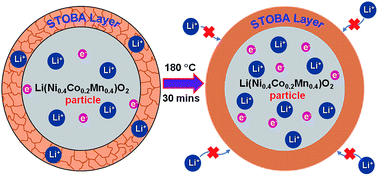
Self-terminated hyper-branched oligomers (STOBA) were coated and then melted on a Li(Ni0.4Co0.2Mn0.4)O2 cathode to form a dense polymer film at high temperatures. The physical and structural changes of the polymer layer at different temperatures and charge conditions were investigated by nitrogen adsorption–desorption, X-ray photoelectron spectroscopy, resistance measurements, scanning electron microscopy, and solid-state 7Li-NMR and 13C-NMR spectroscopy in order to improve the understanding of the role of the STOBA layer in the enhancement of the safety mechanism of lithium ion batteries. The morphological change of the STOBA layer from the porous to nonporous state at the temperature of a thermal runaway of a battery was demonstrated. The change in the resistance values at high temperatures revealed that the STOBA coating is helpful for the prevention of internal short-circuiting and thermal runaway. Most importantly, the 7Li-NMR results acquired at a very high spinning speed (50 kHz) allow the monitoring of the subtle changes in the local environments of the Li+ ions and their interaction and mobility in the STOBA–cathode interface as functions of temperature and charge states. The combined characterization results improve the understanding of how the STOBA layer can contribute to the safety features of lithium ion batteries.


 Please wait while we load your content...
Please wait while we load your content...